 Rivista in lingua italiana
Rivista in lingua italiana
riservata ai Soci SISA
Ultimo numero:
Anno 16 • N.1/2025
SISANews
Il trial CARMELINA: linagliptin ed eventi cardiovascolari maggiori in adulti con diabete di tipo 2
Studi precedenti hanno dimostrato la sicurezza CV di 3 inibitori selettivi della dipeptidil-peptidasi 4 (o gliptine), ma avevano incluso un numero limitato di pazienti con alto rischio CV e malattia renale cronica. CARMELINA è un trial clinico di non inferiorità, randomizzato, controllato con placebo e multicentrico, condotto da agosto 2013 ad agosto 2016 in oltre 600 centri in 27 Paesi, che ha arruolato adulti con diabete di tipo 2 ad alto rischio CV e ad alto rischio di patologie renali. Dai risultati dello studio è emerso che linagliptin non è inferiore al placebo, entrambi aggiunti alla terapia antidiabetica tradizionale, per l'outcome primario dei 3 eventi cardiovascolari maggiori considerati.
![]()
Effect of Linagliptin vs Placebo on Major Cardiovascular Events in Adults With Type 2 Diabetes and High Cardiovascular and Renal Risk: The CARMELINA Randomized Clinical Trial
Rosenstock J, Perkovic V, Johansen OE, Cooper ME, Kahn SE, Marx N, Alexander JH, Pencina M, Toto RD, Wanner C, Zinman B, Woerle HJ, Baanstra D, Pfarr E, Schnaidt S, Meinicke T, George JT, von Eynatten M, McGuire DK; CARMELINA Investigators
JAMA 2019;321:69-79
Importance: Type 2 diabetes is associated with increased cardiovascular (CV) risk. Prior trials have demonstrated CV safety of 3 dipeptidyl peptidase 4 (DPP-4) inhibitors but have included limited numbers of patients with high CV risk and chronic kidney disease. Objective: To evaluate the effect of linagliptin, a selective DPP-4 inhibitor, on CV outcomes and kidney outcomes in patients with type 2 diabetes at high risk of CV and kidney events. Design, Setting, and Participants: Randomized, placebo-controlled, multicenter noninferiority trial conducted from August 2013 to August 2016 at 605 clinic sites in 27 countries among adults with type 2 diabetes, hemoglobin A1c of 6.5% to 10.0%, high CV risk (history of vascular disease and urine-albumin creatinine ratio [UACR] >200 mg/g), and high renal risk (reduced eGFR and micro- or macroalbuminuria). Participants with end-stage renal disease (ESRD) were excluded. Final follow-up occurred on January 18, 2018. Interventions: Patients were randomized to receive linagliptin, 5 mg once daily (n?=?3494), or placebo once daily (n?=?3485) added to usual care. Other glucose-lowering medications or insulin could be added based on clinical need and local clinical guidelines. Main Outcomes and Measures: Primary outcome was time to first occurrence of the composite of CV death, nonfatal myocardial infarction, or nonfatal stroke. Criteria for noninferiority of linagliptin vs placebo was defined by the upper limit of the 2-sided 95% CI for the hazard ratio (HR) of linagliptin relative to placebo being less than 1.3. Secondary outcome was time to first occurrence of adjudicated death due to renal failure, ESRD, or sustained 40% or higher decrease in eGFR from baseline. Results: Of 6991 enrollees, 6979 (mean age, 65.9 years; eGFR, 54.6 mL/min/1.73 m2; 80.1% with UACR >30 mg/g) received at least 1 dose of study medication and 98.7% completed the study. During a median follow-up of 2.2 years, the primary outcome occurred in 434 of 3494 (12.4%) and 420 of 3485 (12.1%) in the linagliptin and placebo groups, respectively, (absolute incidence rate difference, 0.13 [95% CI, -0.63 to 0.90] per 100 person-years) (HR, 1.02; 95% CI, 0.89-1.17; P<0.001 for noninferiority). The kidney outcome occurred in 327 of 3494 (9.4%) and 306 of 3485 (8.8%), respectively (absolute incidence rate difference, 0.22 [95% CI, -0.52 to 0.97] per 100 person-years) (HR, 1.04; 95% CI, 0.89-1.22; P?=?.62). Adverse events occurred in 2697 (77.2%) and 2723 (78.1%) patients in the linagliptin and placebo groups; 1036 (29.7%) and 1024 (29.4%) had 1 or more episodes of hypoglycemia; and there were 9 (0.3%) vs 5 (0.1%) events of adjudication-confirmed acute pancreatitis. Conclusions and Relevance: Among adults with type 2 diabetes and high CV and renal risk, linagliptin added to usual care compared with placebo added to usual care resulted in a noninferior risk of a composite CV outcome over a median 2.2 years. Trial Registration: ClinicalTrials.gov Identifier: NCT01897532..
Area Soci
Eventi
39° Congresso Nazionale
 39° Congresso Nazionale
39° Congresso NazionaleRoma, 23-25 novembre 2025
Save the date




 Spring Meeting Gruppi Giovani SID, SIGG, SIIA, SIMI, SIPREC, SISA
Spring Meeting Gruppi Giovani SID, SIGG, SIIA, SIMI, SIPREC, SISARimini, 6-8 aprile 2025
[continua a leggere]
 SISA LIPID ACADEMY - Corso avanzato di lipidologia clinica
SISA LIPID ACADEMY - Corso avanzato di lipidologia clinicaModena, 4-5 Luglio 2024
[continua a leggere]Giornale Italiano Arteriosclerosi
HoFH today
 Rivista Italiana della
Rivista Italiana della
Ipercolesterolemia
Familiare Omozigote
Anno 6 • N.1/2024
Rivista NMCD
Diateca
[continua a leggere]
[continua a leggere]
Newsletter
il vostro indirizzo di posta elettronica
Progetto LIPIGEN
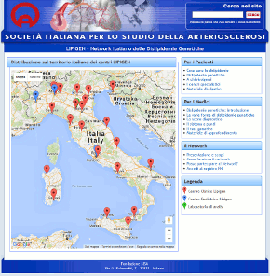
Nuovo sito dedicato al Progetto LIPIGEN
Progetto LIPIGEN - Vecchio portale
E' necessario essere loggati come utente
Lipigen per poter accedere alla pagina
PROject Statin Intolerance SISA
PROSISA – PROject Statin Intolerance SISA
E' necessario essere loggati come utente
PROSISA per poter accedere alla pagina
GILA - Lipoprotein Aferesi
Gruppo Interdisciplinare Lipoprotein Aferesi
(Accesso Gruppo GILA-Lipoprotein Aferesi)
E' necessario essere loggati come utente del Gruppo GILA per poter accedere
Gruppo Interdisciplinare Lipoprotein Aferesi
(Documentazione ad accesso libero)
Pagina informativa per medici e pazienti










