 Rivista in lingua italiana
Rivista in lingua italiana
riservata ai Soci SISA
Ultimo numero:
Anno 16 • N.1/2025
Abstract
Randomized head-to-head comparison of pitavastatin, atorvastatin, and rosuvastatin for safety and efficacy (quantity and quality of LDL): the PATROL trial
Saku K, Zhang B, Noda K.
Circ J 2011;75:1493-505
BACKGROUND: Atorvastatin, rosuvastatin and pitavastatin are available for intensive, aggressive low-density lipoprotein cholesterol (LDL-C)-lowering therapy in clinical practice. The objective of the Randomized Head-to-Head Comparison of Pitavastatin, Atorvastatin, and Rosuvastatin for Safety and Efficacy (Quantity and Quality of LDL) (PATROL) Trial was to compare the safety and efficacy of atorvastatin, rosuvastatin and pitavastatin head to head in patients with hypercholesterolemia. This is the first prospective randomized multi-center trial to compare these strong statins (UMIN Registration No: 000000586).
METHODS AND RESULTS: Patients with risk factors for coronary artery disease and elevated LDL-C levels were randomized to receive atorvastatin (10mg/day), rosuvastatin (2.5mg/day), or pitavastatin (2mg/day) for 16 weeks. Safety was assessed in terms of adverse event rates, including abnormal clinical laboratory variables related to liver and kidney function and skeletal muscle. Efficacy was assessed by the changes in the levels and patterns of lipoproteins. Three hundred and two patients (from 51 centers) were enrolled, and these 3 strong statins equally reduced LDL-C and LDL particles, as well as fast-migrating LDL (modified LDL) by 40-45%. Newly developed pitavastatin was non-inferior to the other 2 statins in lowering LDL-C. There were no differences in the rate of adverse drug reactions among the 3 groups, but HbA(1c) was increased while uric acid was decreased in the atorvastatin and rosuvastatin groups.
CONCLUSIONS: The safety and efficacy of these 3 strong statins are equal. It is suggested that the use of these 3 statins be completely dependent on physician discretion based on patient background.
Area Soci
Eventi
39° Congresso Nazionale
 39° Congresso Nazionale
39° Congresso NazionaleRoma, 23-25 novembre 2025
Save the date




 Spring Meeting Gruppi Giovani SID, SIGG, SIIA, SIMI, SIPREC, SISA
Spring Meeting Gruppi Giovani SID, SIGG, SIIA, SIMI, SIPREC, SISARimini, 6-8 aprile 2025
[continua a leggere]
 SISA LIPID ACADEMY - Corso avanzato di lipidologia clinica
SISA LIPID ACADEMY - Corso avanzato di lipidologia clinicaModena, 4-5 Luglio 2024
[continua a leggere]Giornale Italiano Arteriosclerosi
HoFH today
 Rivista Italiana della
Rivista Italiana della
Ipercolesterolemia
Familiare Omozigote
Anno 6 • N.1/2024
Rivista NMCD
Diateca
[continua a leggere]
[continua a leggere]
Newsletter
il vostro indirizzo di posta elettronica
Progetto LIPIGEN
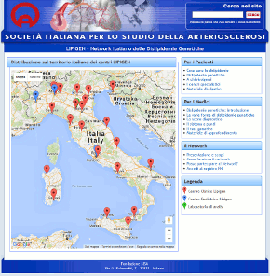
Nuovo sito dedicato al Progetto LIPIGEN
Progetto LIPIGEN - Vecchio portale
E' necessario essere loggati come utente
Lipigen per poter accedere alla pagina
PROject Statin Intolerance SISA
PROSISA – PROject Statin Intolerance SISA
E' necessario essere loggati come utente
PROSISA per poter accedere alla pagina
GILA - Lipoprotein Aferesi
Gruppo Interdisciplinare Lipoprotein Aferesi
(Accesso Gruppo GILA-Lipoprotein Aferesi)
E' necessario essere loggati come utente del Gruppo GILA per poter accedere
Gruppo Interdisciplinare Lipoprotein Aferesi
(Documentazione ad accesso libero)
Pagina informativa per medici e pazienti










